
In 2000, our group published the characterisation of the transcription factor Snail1 as a repressor of E-cadherin expression, a protein key in determining the epithelial phenotype (Batlle et al., Nat Cell Biol 2000, 2:84 -89). Snail1 also induces the conversion of the epithelial cells into mesenchymal cells (epithelial-mesenchymal transition, EMT). This discovery led the group to study this factor and its function as a determinant of the EMT, a transition that not only provides cells with major invasive capacity but also more resistance to apoptosis and even stem cell characteristics. Dr. García de Herreros' group is interested in studying the function of Snail1 in adult animals, with special emphasis on its role in mesenchymal cells and their relationship with neoplasms. In recent years, the group has analysed how Snail1 acts as a repressor in epithelial genes, as well as characterising the mechanism used to activate mesenchymal genes. Other significant results include the characterisation of RNA repeats in heterochromatin, both pericentromeric and telomeric, as a target for Snail1 that must be transiently repressed so that the EMT occurs correctly and the mesenchymal genes are activated. We have also studied how Snail1 expression is controlled, focusing in the role of ubiquitin ligases and deubiquitinases that modulate Snail1 protein half-life. We have recently characterized a new deubiquitinase, Usp27X that antagonizes the action of Snail1 E3 ligases and stabilizes Snail1 during TGFb-induced EMT.
Other group's recent works have assessed the importance of Snail 1 in the process of fibroblast activation. It is becoming increasingly clear that stromal cells play a crucial role in the development and progression of a tumour. Among these cells, fibroblasts seem to be especially significant, and those of the tumour stroma (Cancer-Associated fibroblasts, CAFs) have different characteristics to those of tissues. CAFs are more similar to active fibroblasts characteristic of fibrotic processes. Our results indicate that Snail1 is necessary for the activation of these cells by cytokines such as TGFβ. Fibroblasts that express or do not express Snail1 exhibit a different effect on the growth of tumour cells when both are co-implanted in mice, and they have a differential effect on their ability to invade in vivo and in vitro (Figure 1). According to the results, the elimination of Snail1 in adult mice delays tumour growth in these animals, as well as decrease tumour invasiveness.
The group's current goals are focused on the analysis of the signals produced by active fibroblasts involved in the invasion of epithelial tumour cells, as well as their effects on other cellular components of tumoral stroma. We are also looking at the role of active fibroblasts in other phases of metastasis, such as in the implantation of metastatic cells in target tissues.
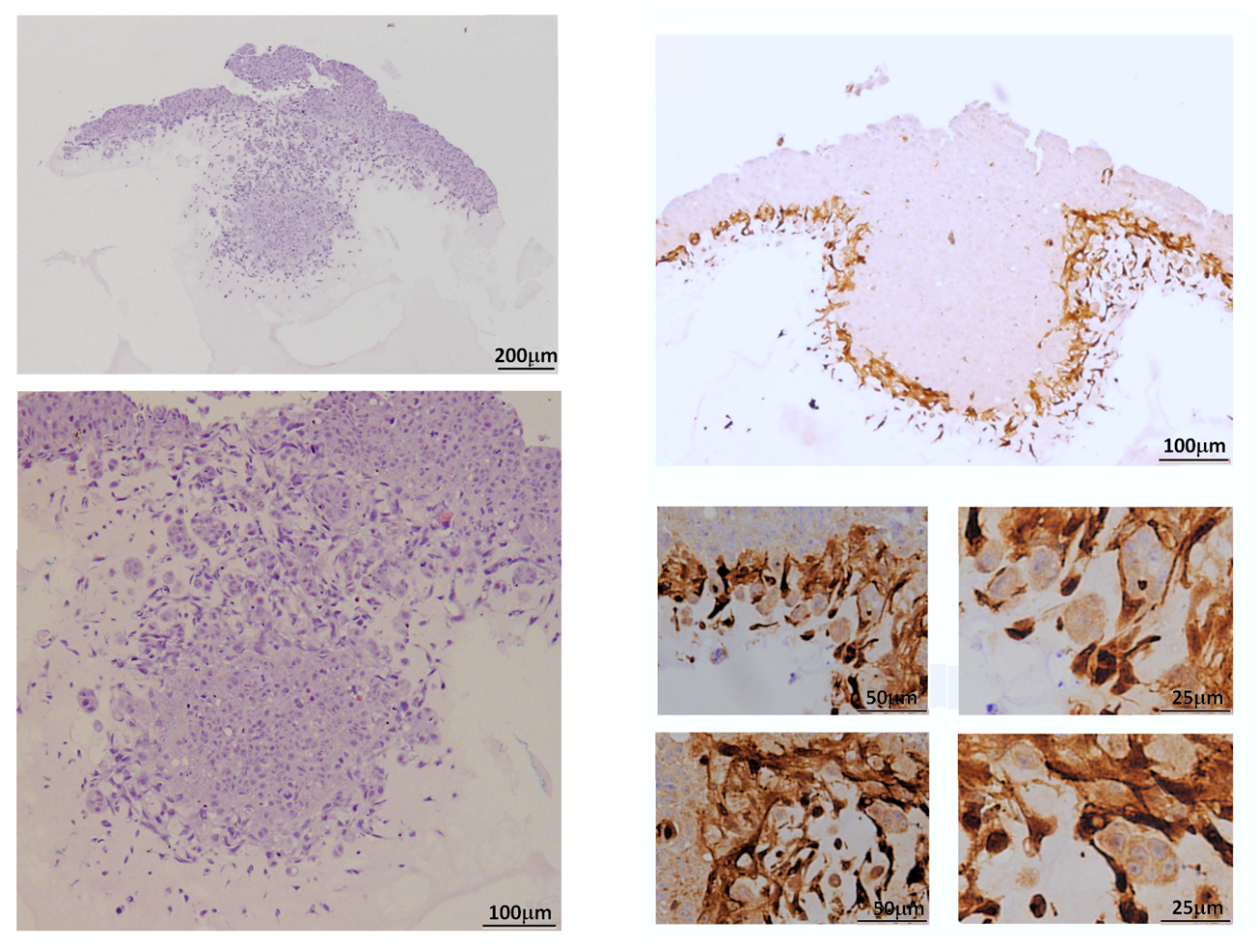
Figure 1: Cooperation of epithelial and mesenchymal cells during the invasion process. HT-29 M6 tumoral epithelial cells were seeded on a matrix of Matrigel/Collagen in the presence of mesenchymal cells marked with GFP. The sections were prepared one week after seeding and dyed with haematoxylin (left) or analysed using immunohistochemistry with an anti-GFP antibody (right). From Alba-Castellón et al, 2016.
Four selected recent publications:
© Institut Hospital del Mar
d'Investigacions MèdiquesLegal Notice and Privacy Policy | Cookie Policy | Site Index | Accessibility | Find Us | Contact